:ouie
Gegenschein
Gegenschein (/ˈɡeɪɡənˌʃaɪn/; German: [ˈɡeːɡn̩ˌʃaɪn]; lit. 'counter-shine') is a faintly bright spot in the night sky centered at the antisolar point. The backscatter of sunlight by interplanetary dust causes this optical phenomenon, also called counterglow.
Gegenschein (/ˈɡeɪɡənˌʃaɪn/; German: [ˈɡeːɡn̩ˌʃaɪn]; lit. 'counter-shine') is a faintly bright spot in the night sky centered at the antisolar point. The backscatter of sunlight by interplanetary dust causes this optical phenomenon, also called counterglow.
Contents
Explanation[edit]
Like zodiacal light, gegenschein is sunlight scattered by interplanetary dust. Most of this dust orbits the Sun near the ecliptic plane, with a possible concentration of particles centered at the L2 point of the Earth–Sun system.[1]
Gegenschein is distinguished from zodiacal light by its high angle of reflection of the incident sunlight on the dust particles. It forms a slightly brighter elliptical spot directly opposite the Sun within the dimmer band of zodiacal light. The intensity of the gegenschein is relatively enhanced because each dust particle is seen at full phase.[2]The Dude Abides
Louis de Broglie
Like zodiacal light, gegenschein is sunlight scattered by interplanetary dust. Most of this dust orbits the Sun near the ecliptic plane, with a possible concentration of particles centered at the L2 point of the Earth–Sun system.[1]
This article needs additional citations for verification. (August 2019) |
Louis de Broglie | |
|---|---|
 Broglie in 1929 | |
| Born | 15 August 1892 Dieppe, France |
| Died | 19 March 1987 (aged 94) Louveciennes, France |
| Nationality | French |
| Alma mater | University of Paris (ΒΑ in History, 1910; BA in Sciences, 1913; PhD in physics, 1924) |
| Known for | Wave nature of electrons De Broglie–Bohm theory de Broglie wavelength |
| Awards | Nobel Prize in Physics (1929) Henri Poincaré Medal (1929) Albert I of Monaco Prize(1932) Max Planck Medal (1938) Kalinga Prize (1952) |
| Scientific career | |
| Fields | Physics |
| Institutions | University of Paris (Sorbonne) |
| Thesis | Recherches sur la théorie des quanta("Research on Quantum Theory") (1924) |
| Doctoral advisor | Paul Langevin |
| Doctoral students | Cécile DeWitt-Morette Bernard d'Espagnat Jean-Pierre Vigier Alexandru Proca Marie-Antoinette Tonnelat |
Louis Victor Pierre Raymond, 7th Duc de Broglie (/də ˈbroʊɡli/,[1] also US: /də broʊˈɡliː, də ˈbrɔɪ/,[2][3] French: [də bʁɔj][4][5] or [də bʁœj] (![]() listen); 15 August 1892 – 19 March 1987)[6] was a French physicist and aristocrat who made groundbreaking contributions to quantum theory. In his 1924 PhD thesis, he postulated the wave nature of electrons and suggested that all matter has wave properties. This concept is known as the de Broglie hypothesis, an example of wave–particle duality, and forms a central part of the theory of quantum mechanics.
listen); 15 August 1892 – 19 March 1987)[6] was a French physicist and aristocrat who made groundbreaking contributions to quantum theory. In his 1924 PhD thesis, he postulated the wave nature of electrons and suggested that all matter has wave properties. This concept is known as the de Broglie hypothesis, an example of wave–particle duality, and forms a central part of the theory of quantum mechanics.
De Broglie won the Nobel Prize for Physics in 1929, after the wave-like behaviour of matter was first experimentally demonstrated in 1927.
The 1925 pilot-wave model,[7] and the wave-like behaviour of particles discovered by de Broglie was used by Erwin Schrödinger in his formulation of wave mechanics.[8] The pilot-wave model and interpretation was then abandoned, in favor of the quantum formalism, until 1952 when it was rediscovered and enhanced by David Bohm.[9]
Louis de Broglie was the sixteenth member elected to occupy seat 1 of the Académie française in 1944, and served as Perpetual Secretary of the French Academy of Sciences.[10][11] De Broglie became the first high-level scientist to call for establishment of a multi-national laboratory, a proposal that led to the establishment of the European Organization for Nuclear Research (CERN).[12]
Biography[edit]
Origin and education[edit]
Louis de Broglie belonged to the famous aristocratic family of Broglie, whose representatives for several centuries occupied important military and political posts in France. The father of the future physicist, Louis-Alphonse-Victor, 5th duc de Broglie, was married to Pauline d’Armaille, the granddaughter of the Napoleonic General Philippe Paul, comte de Ségur and his wife, the biographer, Marie Célestine Amélie d'Armaillé. They had five children; in addition to Louis, these were: Albertina (1872–1946), subsequently the Marquise de Luppé; Maurice (1875–1960), subsequently a famous experimental physicist; Philip (1881–1890), who died two years before the birth of Louis, and Pauline, Comtesse de Pange (1888–1972), subsequently a famous writer.[13] Louis was born in Dieppe, Seine-Maritime. As the youngest child in the family, Louis grew up in relative loneliness, read a lot, and was fond of history, especially political. From early childhood, he had a good memory and could accurately read an excerpt from a theatrical production or give a complete list of ministers of the Third Republic of France. For him it was predicted a great future as a statesman.[14]
De Broglie had intended a career in humanities, and received his first degree in history. Afterwards he turned his attention toward mathematics and physics and received a degree in physics. With the outbreak of the First World War in 1914, he offered his services to the army in the development of radio communications.
Matter wave
| Part of a series of articles about |
| Quantum mechanics |
|---|
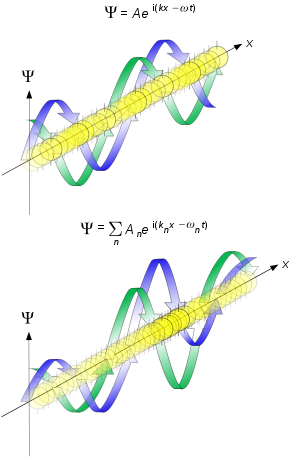
Matter waves are a central part of the theory of quantum mechanics, being an example of wave–particle duality. All matter exhibits wave-like behavior. For example, a beam of electrons can be diffracted just like a beam of light or a water wave. In most cases, however, the wavelength is too small to have a practical impact on day-to-day activities.
The concept that matter behaves like a wave was proposed by French physicist Louis de Broglie (/dəˈbrɔɪ/) in 1924. It is also referred to as the de Broglie hypothesis.[1] Matter waves are referred to as de Broglie waves.
The de Broglie wavelength is the wavelength, λ, associated with a massive particle (i.e., a particle with mass, as opposed to a massless particle) and is related to its momentum, p, through the Planck constant, h:
Wave-like behavior of matter was first experimentally demonstrated by George Paget Thomson's thin metal diffraction experiment,[2] and independently in the Davisson–Germer experiment, both using electrons; and it has also been confirmed for other elementary particles, neutral atoms and even molecules. For its value is the same as the Compton wavelength.
Historical context[edit]
At the end of the 19th century, light was thought to consist of waves of electromagnetic fields which propagated according to Maxwell's equations, while matter was thought to consist of localized particles (see history of wave and particle duality). In 1900, this division was exposed to doubt, when, investigating the theory of black-body radiation, Max Planck proposed that light is emitted in discrete quanta of energy. It was thoroughly challenged in 1905. Extending Planck's investigation in several ways, including its connection with the photoelectric effect, Albert Einstein proposed that light is also propagated and absorbed in quanta; now called photons. These quanta would have an energy given by the Planck–Einstein relation:
and a momentum
where ν (lowercase Greek letter nu) and λ (lowercase Greek letter lambda) denote the frequency and wavelength of the light, c the speed of light, and h the Planck constant.[3] In the modern convention, frequency is symbolized by f as is done in the rest of this article. Einstein's postulate was confirmed experimentally by Robert Millikan and Arthur Compton over the next two decades.
De Broglie hypothesis[edit]
De Broglie, in his 1924 PhD thesis, proposed that just as light has both wave-like and particle-like properties, electrons also have wave-like properties. De Broglie did not simplify his equation into the one that bears his name. He did conclude that hν0 = m0c2.[4][5] He also referred to Einstein’s famous relativity equation. Thus, it was a simple step to get to the equation that bears his name.[6] Also, by rearranging the momentum equation stated in the above section, we find a relationship between the wavelength, λ, associated with an electron and its momentum, p, through the Planck constant, h:[7]
The relationship has since been shown to hold for all types of matter: all matter exhibits properties of both particles and waves.
In 1926, Erwin Schrödinger published an equation describing how a matter wave should evolve – the matter wave analogue of Maxwell's equations — and used it to derive the energy spectrum of hydrogen. Frequencies of solutions of the non-relativistic Schrödinger equation differ from de Broglie waves by the Compton frequency since the energy corresponding to the rest mass of a particle is not part of the non-relativistic Schrödinger equation.
Experimental confirmation[edit]
Matter waves were first experimentally confirmed to occur in George Paget Thomson's cathode ray diffraction experiment[2] and the Davisson-Germer experiment for electrons, and the de Broglie hypothesis has been confirmed for other elementary particles. Furthermore, neutral atoms and even molecules have been shown to be wave-like.
Electrons[edit]
In 1927 at Bell Labs, Clinton Davisson and Lester Germer fired slow-moving electrons at a crystalline nickel target. The angular dependence of the diffracted electron intensity was measured, and was determined to have the same diffraction pattern as those predicted by Bragg for x-rays. At the same time George Paget Thomson at the University of Aberdeen was independently firing electrons at very thin metal foils to demonstrate the same effect.[2] Before the acceptance of the de Broglie hypothesis, diffraction was a property that was thought to be exhibited only by waves. Therefore, the presence of any diffraction effects by matter demonstrated the wave-like nature of matter. When the de Broglie wavelength was inserted into the Bragg condition, the predicted diffraction pattern was observed, thereby experimentally confirming the de Broglie hypothesis for electrons.[9]
This was a pivotal result in the development of quantum mechanics. Just as the photoelectric effect demonstrated the particle nature of light, the Davisson–Germer experiment showed the wave-nature of matter, and completed the theory of wave–particle duality. For physicists this idea was important because it meant that not only could any particle exhibit wave characteristics, but that one could use wave equations to describe phenomena in matter if one used the de Broglie wavelength.
Neutral atoms[edit]
Experiments with Fresnel diffraction[10] and an atomic mirror for specular reflection[11][12] of neutral atoms confirm the application of the de Broglie hypothesis to atoms, i.e. the existence of atomic waves which undergo diffraction, interference and allow quantum reflection by the tails of the attractive potential.[13] Advances in laser cooling have allowed cooling of neutral atoms down to nanokelvin temperatures. At these temperatures, the thermal de Broglie wavelengths come into the micrometre range. Using Bragg diffraction of atoms and a Ramsey interferometry technique, the de Broglie wavelength of cold sodium atoms was explicitly measured and found to be consistent with the temperature measured by a different method.[14]
This effect has been used to demonstrate atomic holography, and it may allow the construction of an atom probe imaging system with nanometer resolution.[15][16] The description of these phenomena is based on the wave properties of neutral atoms, confirming the de Broglie hypothesis.
The effect has also been used to explain the spatial version of the quantum Zeno effect, in which an otherwise unstable object may be stabilised by rapidly repeated observations.[12]
Molecules[edit]
Recent experiments even confirm the relations for molecules and even macromolecules that otherwise might be supposed too large to undergo quantum mechanical effects. In 1999, a research team in Vienna demonstrated diffraction for molecules as large as fullerenes.[17] The researchers calculated a De Broglie wavelength of the most probable C60 velocity as 2.5 pm. More recent experiments prove the quantum nature of molecules made of 810 atoms and with a mass of 10,123 u.[18] As of 2019, this has been pushed to molecules of 25,000 u.[19]
Still one step further than Louis de Broglie go theories which in quantum mechanics eliminate the concept of a pointlike classical particle and explain the observed facts by means of wavepackets of matter waves alone.[20][21][22][23]
De Broglie relations[edit]
The de Broglie equations relate the wavelength λ to the momentum p, and frequency f to the total energy E of a free particle:[24]
where h is the Planck constant. The equations can also be written as
or [25]
where ħ = h/2π is the reduced Planck constant, k is the wave vector, β is the phase constant, and ω is the angular frequency.
In each pair, the second equation is also referred to as the Planck–Einstein relation, since it was also proposed by Planck and Einstein.















Comments
Post a Comment
Have something to say? Say it See it Feel it Live it then take It apart until you know what It is and As you were before you were born, feel the sound of it rattling around in the outbox smell the sight of it as the juices in your brain bag are fed little light waves of electric combinatory discombinatory salty sea solution sliding stealthily supplying the juice for the other end of the switch turned on by the turn on that turns on the on like one is when one is on and one is when one is idling on as the slumber soothes silky roads of well traversed travelers traipsing trails of tuned toned teeny tiny twirls of primes pumping nines into sixes and threes like humming honey bees evens evening out the odds as the unknown unknowns puddle sing a shiny bling and bring the ear funnel Z as close as eyeball close can see...z.z..z...